Frequently Asked Questions
General FAQs
Honey and acacia gum are used in our tablet manufacturing process. Cellulose is often used to give body to the powders. Calcium stearate, derived from vegetable sources, is used as a lubricant to help products release from the tooling of the tablet and capsuling machines.
The calcium stearate we use is derived from palm oil, which is a saturated fat like cocoa butter, dairy fats, etc. It is approved by the FDA to be used as a lubricant and is approved for use in food products.
Many of the ingredients we use in our formulas are plants. With natural variations in plants from harvest-to-harvest there will be changes in color, texture, and odor. The basic formulas remain constant, and quality is ensured by repeat testing.
The calcium stearate we use is derived from palm oil, which is a saturated fat, like cocoa butter, dairy fats, etc. It is approved by the FDA to be used as a lubricant and is approved for use in food products.
Yes. The gelatin is derived from bovine and/or porcine sources. Cellulose is used to make vegetarian capsules.
Our gelatin capsules are derived from bovine and porcine sourced collagen. Capsules used for vegetarian products are made from vegetarian cellulose.
In many cases, yes. However, Zypan, Betaine Hydrochloride, and Cal-Amo should never be chewed. Chlorophyll Complex softgels will temporarily stain the teeth and turn the tongue green. All MediHerb supplements that are labeled as “enteric-coated” should not be chewed.
Some Standard Process supplements are designed as chewables for those who cannot swallow capsules or tablets. If you have difficulty swallowing products that only come in capsules or tablets, we suggest you use a food-safe and clean pill crusher.
For products in a capsule, you can remove the powder from the capsule if you prefer.
Our products are processed in a facility that manufactures other products containing soy, milk, eggs, wheat, peanuts, tree nuts, fish, sesame and shellfish. Please read our labels carefully for any possible ingredient allergens before recommending or consuming supplements for those who are sensitive to certain ingredients. Click here to see our supplements that support the needs of those with dietary restrictions.
Peanuts
Only Peanut Butter Standard Bars continue to be made with peanut butter in a special production suite that is isolated from the rest of the facility. Recognizing the severity of peanut allergies, Standard Process has removed peanut bran from its products and replaced it with rice bran.
Gluten
We follow the FDA guidelines, which state that products that contain less than 20 parts per million (ppm) of gluten can be labeled as gluten-free. All of our products that are designated as gluten-free contain less than 20 ppm and are tested every time they are manufactured to be sure that they meet this standard.
Shellfish
Nutrimere and Iodomere contain New Zealand green mussel. Glucosamine Synergy contains glucosamine sulfate that is derived from crab and shrimp. While not a shellfish, calamari (the source of oil in Calamari Omega-3 Liquid) is in the same family and may trigger a reaction in those who are allergic to shellfish.
Soy
Some products contain soybean ingredients, such as soy powder, soy nuggets, and soy protein. Soybean lecithin, a source of choline and B vitamins, is found in other Standard Process products. It acts as an emulsifier, keeping water and fats from separating in foods. Individual sensitivities to soy protein should be discussed with your practitioner.
We use nutritional yeast, chromium yeast, selenium yeast, and functional yeasts.
Nutritional yeast is a source of certain B vitamins, Chromium yeast is a source of the mineral chromium, selenium yeast is a source of the mineral selenium. Saccharomyces boulardii is a yeast that acts as a type of probiotic.
For over 90 years, Standard Process has been committed to making high quality whole food-based supplements that change lives. Rather than an expiration date, our products use a “Best Buy” date to guarantee product potency. Please refer to the “Best By” date on all product boxes and bottles to determine how long a product maintains optimal quality and/or flavor. Standard Process only guarantees product potency through the “Best By” date. Due to varying degrees of potency, quality and/or flavor of product ingredients overtime, the “Best By” date continues to serve as the best reference for customers.
The shelf life of the product stays the same whether it is opened or unopened.
Below is an example of the different parts of our label and what they refer to.
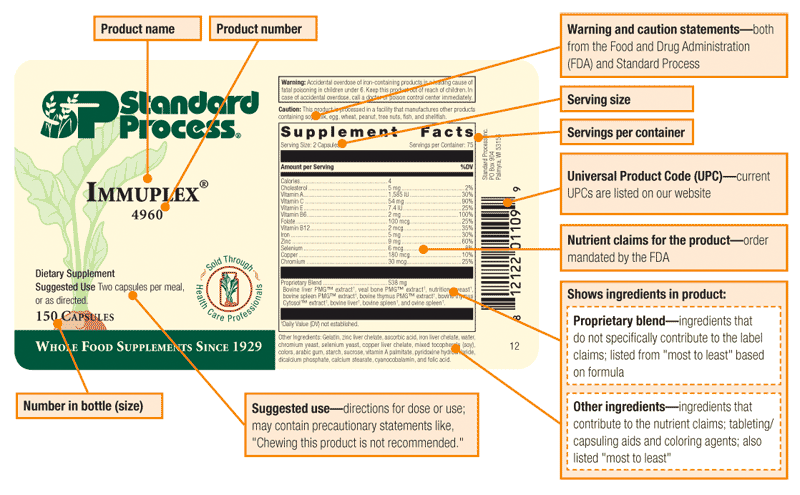
The sucrose found in our products comes from natural sources such as beets and sugar cane. Sucrose, along with starch and acacia gum, is used to coat vitamin A and vitamin D (cholecalciferol), making them into a powdered form that helps prevent oxidation of these vitamins. The amount of sucrose in the actual product is very minimal.
The body processes most compounds based on concentration; so the more there is, the faster your body works to process it. The key is getting the right concentration at the right time. Taking smaller amounts throughout the day allows the body to maintain a lower but more consistent amount for a longer period of time. It is more efficient for your body and more effective in sustaining the molecular mechanisms supported by the compounds in our supplements.
Consistent with our commitment to produce and sell only the highest-quality dietary supplements, each batch of marine oil used in our fish oil products is tested for mercury, potential oxidation, , polychlorinated biphenyls (PCBs), and dioxin. The product is also tested for bacteria and to verify our label claims. These tests are carried out by Standard Process and third-party labs. We use this process for testing Tuna Omega-3 Oil, Tuna Omega-3 Chewable, Calamari Omega-3 Liquid, and Cod Liver Oil. We also use this process for testing Olprima™ EPA, Olprima™ DHA, and Olprima™ EPA|DHA.
During manufacturing, our marine oils are processed to retain their natural triglyceride structure.
We also take great care in sourcing our marine-oil products. Tuna comes from the abundant skipjack and/or yellowfin tuna populations. Our calamari oil is certified as sustainable by a nonprofit, nongovernmental organization, Friend of the Sea®.
No, Standard Process does not use any genetically modified organisms. Standard Process is committed to the health-giving properties of whole food nutrition and using ingredients that have not been genetically modified. As part of this commitment, we first source ingredients from our own 868-acre certified organic farm. Certified organic farming does not allow for genetic engineering. A great majority of all raw plant ingredients used to make our products are grown on our own organic farm. When sourcing ingredients we can’t grow on our farm, we are committed to selecting partners who offer ingredients that have not been genetically engineered. We rigorously evaluate all outside vendors and validate the raw ingredients they supply.
In 2016, label requirements changed for how some nutrient measurements are listed. Rather than measurements listed in IU many are now listed in mcg. See chart for sample conversions:
| Vitamin A/Beta-carotene | |
| To convert from IU of vitamin A or beta-carotene to mcg RAE (Retinol Activity Equivalents), multiply by 0.3 | |
| Previous Value | Updated Equivalent |
|---|---|
| 667 IU | 200 mcg |
| 1,667 IU | 500 mcg |
| 3,333 IU | 1,000 mcg |
| 6,667 IU | 2,000 mcg |
| Vitamin D3 | |
| To convert from IU of vitamin D3 to mcg of vitamin D3, multiply by 0.025 | |
| Previous Value | Updated Equivalent |
|---|---|
| 100 IU | 2.5 mcg |
| 200 IU | 5 mcg |
| 400 IU | 10 mcg |
| 800 IU | 20 mcg |
| 2,000 IU | 50 mcg |
| 4,000 IU | 100 mcg |
| Vitamin E | |
| To convert from IU of vitamin E to mg of d-alpha tocopherol, multiply by 0.67 | |
| Previous Value | Updated Equivalent |
|---|---|
| 75 IU | 50 mg |
| 149 IU | 100 mg |
| Folate | |
| To convert from mcg DFE (Dietary Folate Equivalents), divide by 0.6. This conversion applies to both folic acid and 5-MTHF | |
| Previous Value | Updated Equivalent |
|---|---|
| 400 IU | 667 mcg |
| 800 IU | 1,333 mg |
By following the Standard Process Purification Program, you will be working toward sustained, long-term weight management. The ideal weight-management system is not a quick fix but a long-term commitment. You will need to make lifestyle changes that will continue long after the purification process has been completed.
Exercise facilitates the removal of toxins and helps maintain a healthy weight. It is recommended that you walk 30-45 minutes at least four days per week. Strenuous exercise should be put on hold during the 21-day program. Consult your health care professional if you are currently on a more strenuous exercise program that you would like to maintain throughout the purification program or about resuming strenuous exercise post-purification.
Here are some tips to help you stay on track:
- Plan your strategy before you leave home.
- When choosing a restaurant, select one that serves foods on the recommended list. This will curb temptation
- When traveling, bring along a cooler packed with water bottles, fresh fruit, and sliced raw vegetables.
- When attending a social gathering, bring a dish to share that fits your stage of the program, like a fruit/veggie tray or chicken kabobs.
Sign up to receive daily encouraging emails and an invitation to our exclusive Facebook group for purification program participants. (Link for Facebook)
Based on your health history and goals, your health care professional can help you decide if you should continue taking medications or other supplements during this program. They might also recommend additional supplements to support your body while on the program.
These vegetables were selected for their high nutritive value and their capacity to support purification. Other vegetables can be consumed in moderation, however do not consume corn or white potatoes.
It is normal for participants to experience some fatigue or lack of energy when beginning the purification program. You may be fatigued because your body may require a higher amount of protein, water, or exercise during purification and post-purification. Make sure you are prioritizing sleep. Ask your health care professional about adding more protein to your diet. Veg-E Pro Complete or Whey Pro would be good options.
Remember to drink plenty of water, at least 64 ounces per day. Eat plenty of vegetables and fruits with a high fiber content. Eating one beet daily encourages regular bowel movements.
Protein is important for supporting detoxification processes. Whey is a protein source found in SP Complete, and lactose is a nutrient found in the whey. There is approximately ½ gram of lactose per shake. Try SP Complete Dairy Free if lactose is a concern.
The shakes and supplements you will take are vegetarian (lacto-ovo) but not vegan. You can make it vegan by choosing SP Complete Diary Free and vegan sources of protein.
If you suspect you are having a reaction to a specific food, refrain from eating it and consult your health care professional about how to incorporate alternative food sources.
Omega-3s, short for omega-3 fatty acids, is a family of essential fatty acids that play important roles in the human body. Since EPA and DHA are not made efficiently by the body, it is recommended that these be consumed at higher levels in the diet.
Omega-3 fatty acids are polyunsaturated fatty acids (PUFAs), meaning that they have several double bonds in the chemical structure. Omega-3s include alpha-linolenic acid (ALA), docosahexaenoic acid (DHA), Eicosapentaenoic acid (EPA) and the less known docosapentaenoic acid (DPA). ALA mainly is found in plants, while DHA and EPA are mainly found in animal foods and algae.
EPA is associated with a positive effect on emotional balance. Just fifteen percent of ALA turns into EPA, and even a smaller amount is eventually converted to DHA. Both EPA and DHA are precursors for pro-resolving mediator that biosynthesized to resolvins, protectins, and maresins which are named for their role in resolving the inflammatory response.
DHA is the most abundant omega-3 in the brain. It is a major component of the neuronal membrane and plays an important role in normal brain structure. Both EPA and DHA are required for manufacturing substances (resolvins, protectins, and maresins) involved in resolving inflammation in the body.
Omega-3 fatty acids play critical roles in the human body and are abundant in the brain and retina. They act as components of the phospholipid structures of cell membrane, play a role in signal transduction and serve as an energy source. Omega-3 fatty acids and their metabolites regulate various functions in the body including cardiovascular, pulmonary, immune, inflammation and the endocrine system.
The Standard American Diet (SAD) lacks the nutritional value that is necessary for living a healthy lifestyle. The human body needs fatty acids found in both omega-3 and omega-6, but it is important that they be consumed in a healthy ratio. The SAD has an overabundance of omega-6 fatty acids, found in poultry, eggs and vegetable oils, and an insufficient amount of omega-3 fatty acids. High omega-6 and low omega-3 consumption can tip a healthy body system out of balance and allow low levels of inflammation to occur. The body needs a higher ratio of omega-3 to omega-6 to support the resolution phase of the inflammation process.
Omega-3s play critical roles in the human body and are abundant in the brain and retina. These essential fatty acids and their metabolites regulate a number of body systems including cardiovascular, pulmonary, immune, nervous, and endocrine systems. Omega-3s are components of the phospholipid structures of the cell membrane and are important for cell membrane structure stability and fluidity. Phospholipids are also a good source of energy and play a role in cell signaling. In short, like oil for any machine, omega-3s help the body operate smoothly.
Omega-3 fatty acids are needed for optimal function of the human body, and they may also provide numerous powerful health benefits. Scientific evidence from several clinical intervention studies has shown that adequate intake of omega-3 fatty acids is associated with several beneficial health outcomes. An emerging number of clinical studies have investigated the actions of EPA, DPA and DHA individually rather than together. It is now possible to tailor different blends of individual omega-3s, mainly EPA and DHA, according to the health benefits we are seeking to achieve.
Also derived from hemp, CBD oil products are typically formulated to deliver a stated amount of CBD. Many states have passed laws permitting the sale and use of CBD oil for specific circumstances, often restricting use for certain medical conditions, and requiring supervision by specific medical practitioners. Our hemp oil products are not formulated to deliver any specific amount of CBD, but contains 15 mg of phytocannabinoids per serving.
Our hemp oil products are safe, effective products for individuals. Standard Process proudly grows hemp on our certified organic farm, which helps us preserve its exceptional quality from seed to supplement.
FDA strongly advises that during pregnancy or while breastfeeding, you avoid using cannabis (hemp) or cannabis-derived products, CBD, THC, or marijuana in any form. Do not put yourself or your baby at risk by using cannabis products while pregnant or breastfeeding.
The proper functioning of the ECS, the body’s primary regulatory system is known as endocannabinoid tone. A variety of factors can influence the ECS and support its tone, including healthy lifestyle habits, such as diet, exercise, stress modification, acupuncture, massage and using dietary supplements.
The Endocannabinoid System (ECS) and a healthy diet may support key resolution pathways to inflammation, energy balance and metabolism through nutrient-rich omega-3 fatty acids, antioxidants, and fibers from a whole food matrix in the form of phytonutrients.
A large percentage of the U.S. adult population falls below the recommendations for omega-3 fatty acid consumption, including ALA, EPA, and DHA, which are required for normal systemic body functions and must be obtained from dietary sources. Omega-3 and omega-6 essential fatty acids are precursors of specialized lipid mediators (eicosanoids), which play an important role in the regulation of inflammation and the immune response. A balanced combination of omega-3 and omega-6 fatty acids favors healthy inflammation processes and supports the resolution phase of the inflammation processes by regulating various systemic functions in the body, including cardiovascular, pulmonary, immune, and endocrine systems.
Hemp Oil Complex provides three benefits in one product. TrueBroc® contains broccoli, and cruciferous vegetables like broccoli are rich in sulfur-containing compounds called glucosinolates. Glucoraphanin, the type of glucosinolate in broccoli, helps to form sulforaphane, a biologically active isothiocyanate. The biological actions of glucosinolate-derived isothiocyanates with active myrosinase enhance the bioavailability of isothiocyanates thereby supporting cellular antioxidant capacity. Studies have shown that many isothiocyanates found in plants such as broccoli, particularly sulforaphane, increased expression of antioxidant enzymes via the activation of the nuclear factor-2 (Nrf-2) dependent pathway.
Standard Process products are thought to work synergistically and support multiple aspects of health. In some cases, combined products can result in very high levels of certain vitamins or minerals. Where warranted, guidance is provided for individual products on the label and in this catalog via warnings and cautions. Additional information about vitamin and mineral levels is provided through the “daily values” reported on the label. These values, set by the Food and Drug Administration, indicate how much of the recommended daily consumption of the nutrient is found in a serving. Several excellent resources detailing vitamin/mineral action and current knowledge regarding recommended levels are available online.
- Click on the “Sign-In” button at the top of your screen.
- Once you are logged in, you are able to find educational events on the bottom of our main page.
Recorded webinars can be accessed by clicking on “Resources” in the main navigation and then selecting “Education on Demand”.
Maltodextrin is used in very small quantities to ensure even distribution and potency of ingredients and helps with tableting and flow. It is a purified carbohydrate, but does not make a significant contribution.
Side effects could vary based on your product selection and individual health needs. Please consult with your health care professional for more information.
Our products are best when stored in a cool, dry place, away from direct sunlight.
For information or advice on which supplements to take or how to take them, please contact your health care provider.
Order Questions
At Standard Process, we believe your unique nutritional requirements are best evaluated by a consultation with a health care practitioner. They will ensure you have the right products to meet the needs of you and your family, including your pets. Contact one of our caring, qualified practitioners to get started with our products.
- Visit a practitioner in person to purchase at their office. Click here to find a practitioner near you. Enter your zip code and select “In Office” under “Purchasing Options.”
- Order from one of our practitioners online. Click here to find a practitioner near you. Enter your zip code and select “Online” under “Purchasing Options.”
- Some of our practitioners offer the option to purchase online after an in-person consultation. After consultation, they can determine the best product array for your health needs. For a list of practitioners who offer this service, Click here to enter your zip code and select “Patient Direct” under “Purchasing Options.”
- We offer a select number of our products on Amazon through our authorized reseller, Pattern/iServe.
We work with both UPS and FedEx, and we select our shipping partner based on fastest delivery time to your location.
- Orders placed Monday – Friday before 3 p.m. CT usually ship the same day (expect on major holidays). Orders placed on Friday after 3 pm central will ship the following Monday. Our typical delivery time is one to five business days after ordering.
When you place your order, an estimated delivery date and shipping options/costs will be provided.
If there is no one to receive/sign for the package(s) on the first delivery, UPS will make two additional attempts to deliver, after which the package(s) will be returned to Standard Process. If returned, the package(s) will be subject to a 20% charge plus any additional shipping fees.
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
This information helps us understand your needs better and enables us to assist you promptly and effectively.
If the return request is due to a quality issue, please include pictures.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Amazon Purchaser- You will need to contact our authorized 3rd Party seller, Pattern. (Customer Service Phone: 866-765-1355), or if purchased through another seller, contact the seller directly to initiate any returns or resolve any issues with your order. As the seller manages transactions and returns on their platform, they will be best equipped to assist you promptly.
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
This information helps us understand your needs better and enables us to assist you promptly and effectively.
If the return request is due to a quality issue, please include pictures.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Amazon Purchaser-You will need to contact our authorized 3rd Party seller, Pattern. (Customer Service Phone: 866-765-1355), or if purchased through another seller, contact the seller directly to initiate any returns or resolve any issues with your order. As the seller manages transactions and returns on their platform, they will be best equipped to assist you promptly.
If you receive packages in a damaged condition from Standard Process, it’s crucial to report it promptly to ensure proper resolution. Here’s what you should do:
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
Please include pictures when reaching out regarding broken or damaged bottles.
We ask that you save all damaged products, packaging material, and shipping boxes until a resolution is reached.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
Please include pictures when reaching out regarding a quality issue.
This information helps us understand your needs better and enables us to assist you promptly and effectively.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
This information helps us understand your needs better and enables us to assist you promptly and effectively.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Amazon Purchaser-You will need to contact our authorized 3rd Party seller, Pattern. (Customer Service Phone: 866-765-1355), or if purchased through another seller, contact the seller directly to initiate any returns or resolve any issues with your order. As the seller manages transactions and returns on their platform, they will be best equipped to assist you promptly.
In general, Standard Process does not accept expired product back for an exchange or refund if its past its expiration date.
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
This information helps us understand your needs better and enables us to assist you promptly and effectively.
If the return request is due to a quality issue, please include pictures.
Customer Care Department: 800-558-8740
Email: sporders@standardprocess.com
Amazon Purchaser-You will need to contact our authorized 3rd Party seller, Pattern. (Customer Service Phone: 866-765-1355), or if purchased through another seller, contact the seller directly to initiate any returns or resolve any issues with your order. As the seller manages transactions and returns on their platform, they will be best equipped to assist you promptly.
Manufacturing Questions
Honey, acacia gum, and cellulose are used as binders.
Honey, acacia gum, cellulose gum, and cellulose are used as binders. Calcium stearate and calcium lactate, derived from vegetable sources, is used as a lubricant to help the products flow into the tablet machines.
The supplement industry is regulated and inspected by various governmental agencies. Standard Process Inc. is regulated and inspected by:
- The Food and Drug Administration (FDA)
- The United States Department of Agriculture (USDA)
- Wisconsin Department of Agriculture (WI-DA)
- Occupational Safety and Health Administration (OSHA)
- Midwest Organic Services Association (MOSA)
Wisconsin Department of Natural Resources (DNR)
Between each separate product run, any equipment that comes in contact with ingredients or finished product is disassembled and washed.
- Cleaning agents are rotated monthly to avoid bacterial resistance.
- Swab samples are taken from several areas of each piece of equipment. These samples are exposed to a luminometer, which fluoresces in proportion to the amount of adenosine triphosphate (ATP) in the sample. ATP is found in and around all biological matter and would indicate a possible bacterial presence.
- If the luminometer measures ATP results beyond an acceptable level, the cleaning process is repeated and additional ATP tests are conducted.
- This is done as many times as needed to obtain acceptable samples.
- After the cleaned equipment passes the ATP testing protocol, the equipment is sanitized. Only then is the equipment reassembled for use with a new batch of product.
- If a clean piece of equipment is unused for more than 24 hours, it is re-sanitized prior to use.
- In addition to the equipment itself, the surrounding production suite, including the floors, walls, and vent covers, is cleaned.
All raw animal tissues are USDA-approved, from facilities inspected by the USDA or the Canadian Food Inspection Agency (CFIA). All organs are inspected for safety, contaminants, and are received with a Certificate of Analysis.
For additional information, click on the following government resource:
Standard Process has a number of hemp oil products for both adults (people over the age of 17) and pets (dogs and cats). People and pets outside those parameters should not consume our hemp oil products.
We test all products for heavy metals contamination, which includes arsenic, mercury.
Farm Questions
Currently, Standard Process has 868 acres of organically certified farm land used to grow vegetables and other plant ingredients for our supplements. We grow anywhere from 12 to 24 different crops and variations in a single year. Our crops include: alfalfa, barley grass, beets, broccoli, Brussels sprouts, buckwheat, carrots, hemp, kale, kidney beans, oats, pea vine, and Spanish black radish, as well as a variety of cover crops.
We do not house or raise any animals on our farm, though our farm is home to a number of pollinator species . Read more about our certified organic farm here .
When soil tests show a need for a nutrient, we apply organic fertilizers that are included in the OMRI (Organic Materials Review Institute) list of approved materials. We also rotate crops extensively to ensure we have a variety of residues in our soil. We believe in the concept of green manure, and we often have 1/3 of the Standard Process acres in some form of green manure rotation at all times.
Patient Direct
Patient Direct is an online ordering platform that helps you connect your current patients, under your supervision, with the Standard Process and MediHerb® nutritional supplements they need. It offers an efficient way to offer online ordering without any additional technology or training. With your approved account, you will also have access to marketing tools, reporting, and resources to support your business. All of the inventory, logistics, and technical requirements are managed for you by Standard Process.
Patient Direct is available to most health care professionals who have a Standard Process account in good standing. If you are interested in registering for a Patient Direct account, you can click here.
Current patients of participating health care professionals may register here with their practitioner’s unique Patient Direct code. You will need this code to start your application.
If your health care professional does not currently participate in the program, click here to find an HCP who does participate.
Patient Direct offers an efficient way for your practitioner to select and recommend supplements that are specific to your personal nutritional needs. Once you have registered for a Patient Direct account, using your practitioner’s unique access code, they will approve you within their practitioner portal. Then they will be able to add the Standard Process and MediHerb® products that best fit your needs. When you order using Patient Direct, the products will ship directly from the Standard Process facility to your doorstep.
We also offer auto-delivery and easy reorder capabilities on Patient Direct orders.
If you don’t have a health care professional, please visit our “Where To Buy” page or contact our Customer Care department at 800-558-8740 for assistance in finding a health care professional.
- Ask your health care professional for the Patient Direct Code.
- Go to https://my.standardprocess.com/Patient-Direct
- Scroll down and click the “Register” button in the patient area.
- Read the terms of use and enter the six-digit code provided by your health care professional. Do not share this code with anyone.
- Complete the 3-step registration. Upon completion an email will be sent to your health care professional for approval.
- Your health care professional will act upon your registration; you will receive an email that your account is approved.
- Click on the link inside the email to complete your Standard Process website membership. You must create a username and password.
- Log in to your Patient Direct account at standardprocess.com and begin ordering.
Patient Direct is available to most health care professionals who have a Standard Process account in good standing. If you are interested in applying for a Patient Direct account, you can click here.
We ship to all 50 states as well as Guam, Puerto Rico and the Virgin Islands. Orders of $149.99 or less have a flat rate shipping rate of $12.00 regardless of size, weight, or distance. Standard shipping is free for orders of $150.00 and greater. We offer expedited shipping for an additional charge. Expedited options are displayed during the checkout process and the rate will be applied automatically to orders upon checkout.
Our shipping department hours are Monday-Friday 8am – 3pm CST (excluding major holidays). Orders will typically ship within one to three business days.
You can check your email for the shipping confirmation, which includes tracking information. Or log in to your Patient Direct account at standardprocess.com and select “View Previous Orders” from the drop-down menu under “Patient Direct.” Or, “View Previous Orders” can be found on your “My Account” page under “Order Management”.
Call your health care professional with any questions about supplements.
Contact a Standard Process customer care representative at 800-558-8740 or email patientdirect@standardprocess.com.
Health care professionals are welcome to schedule a tour. Please call 800-848-5061 for more information.
Put simply, we believe the relationship between health care professionals and patients is critical in supporting the best health outcomes.
You know the nutritional needs of your patients and understand Standard Process and MediHerb® products, enabling you to provide guidance, recommendations, and support.
Please contact us within 30 days from your order date to receive assistance.
When contacting us please provide relevant details such as your:
- Account Number
- Order Number
- Product Number
- Lot Number
This information helps us understand your needs better and enables us to assist you promptly and effectively.
If the return request is due to a quality issue, please include pictures.
Customer Care Department: 800-558-8740
Your remuneration will be adjusted based on the amount of the returned product.
Email: sporders@standardprocess.com
Please contact Customer Care to make changes to your primary changes.
1. Visit my.standardprocess.com/account
2. Click on your name to access the My Account page
3. Click on the auto-delivery link on the right side of the screen
4. From the My Auto Deliveries page, you can edit an auto-delivery frequency, quality, content, next ship date, address or payment option
5. Click the edit icon in the upper right corner of each product you would like to manage
There could be multiple reasons why an account may have a hold on it. Please contact Customer Care at (800-848-5061) or at sporders@standardprocess.com.
Standard Process offers accounts to qualified Health Care Practitioners. To apply, go to https://www.standardprocess.com/Register/Standard-Process-Account
For questions about an existing application Please contact our new accounts team at newaccountrep@standardprocess.com with any questions.
Please refer back to your health care professional or log in to your account to view the product list for pricing.
For most updated release date information, log in to my.standardprocess.com and view the product page.
Our products are not licensed for sale outside of the U.S. We currently ship to all U.S. states, Puerto Rico, U.S. Virgin Islands, Guam and military APO and FPO addresses.








